 Increased ROS levels can decrease sperm motility, DNA integrity and spermatozoa membrane integrity due to lipid peroxidation and may cause the apoptosis of spermatozoa. The presence of such contaminants can lead to increased ROS content, changes in osmolality and pH balance, and negatively affect semen quality. Therefore, centrifugation of canine semen prior to cooled shipment or cryopreservation is routinely performed in clinical practice to remove the potentially harmful effects of prostatic fluid or other contaminants such as urine. Additionally, the admixture of prostatic fluid and other seminal plasma factors in raw samples produces reactive oxygen species (ROS), which are known to be harmful to spermatozoa. These include routine processing methods that can negatively impact the viability, motility, and fertilizing capacity of the sperm cells. However, the gains may be countered by other biologic factors and handling conditions, which can reduce the quality of shipped cooled semen at the time of insemination. Comparable pregnancy rates achieved by cooled and fresh semen inseminations have added to the increase in popularity. The gain in popularity for shipping cooled canine semen is due to the relative ease, decreased expense in logistics compared to frozen semen, and the reduced strain for transporting live animals. The demand for insemination in dogs has significantly increased over the last few decades. The use of cooled and frozen semen for artificial insemination (AI) for canine breeding is vital for overcoming geographic and temporal barriers. In conclusion, our study showed that centrifugation within a range of 400 g–900 g for 5–10 min is appropriate for processing canine semen. Similarly, total and progressive motility did not differ across treatments but declined in all groups from T1 to T3 ( p ≤ 0.02). Spermatozoa membrane integrity was not different between centrifugation groups at any time point ( p ≥ 0.38) but declined significantly during cooling (T1 vs. Sperm losses were minimal, and RRs were similar across treatment groups (median >98%, p ≥ 0.062). Sperm RR (%) was calculated post-centrifugation, and plasma membrane integrity (%, Nucleocounter ® SP-100™), total and progressive motility (%, subjective and computer-assisted sperm analysis), and morphology (%, eosin-nigrosin staining) were assessed on initial raw semen (T0), post-centrifugation (T1), and 24 h (T2) and 48 h (T3) after cooling. Individual ejaculates collected from 14 healthy dogs were split into six treatment groups (400 g, 720 g, and 900 g for 5 or 10 min). Cooled storage under standard shipping conditions was used as a stressor to evaluate long-term treatment effects. We hypothesized that higher gravitational (g) force and longer time of centrifugation would result in improved spermatozoa recovery rate (RR) but poorer semen quality. We aim for NEXT-DAY DELIVERY FOR ALL ORDERS PLACED BEFORE 1PM Due to Covid19 we cannot.Our objective was to determine a clinically relevant range of centrifugation parameters for processing canine semen. With our comprehensive range of products and services for artificial insemination, embryo transfer and related biotechnologies, we effectively support our customers in animal breeding, veterinary medicine and research worldwide. 
Minitube is the world-wide leading supplier of systems for the field of assisted animal reproduction. Breeder's Choice Minitube CushionFluid for Centrifugation - Minitube CushionFluid CushionFluid for centrifugation of equine semen. Using the semen extender will maintain a higher percent of forward motility over time as compared to raw semen. 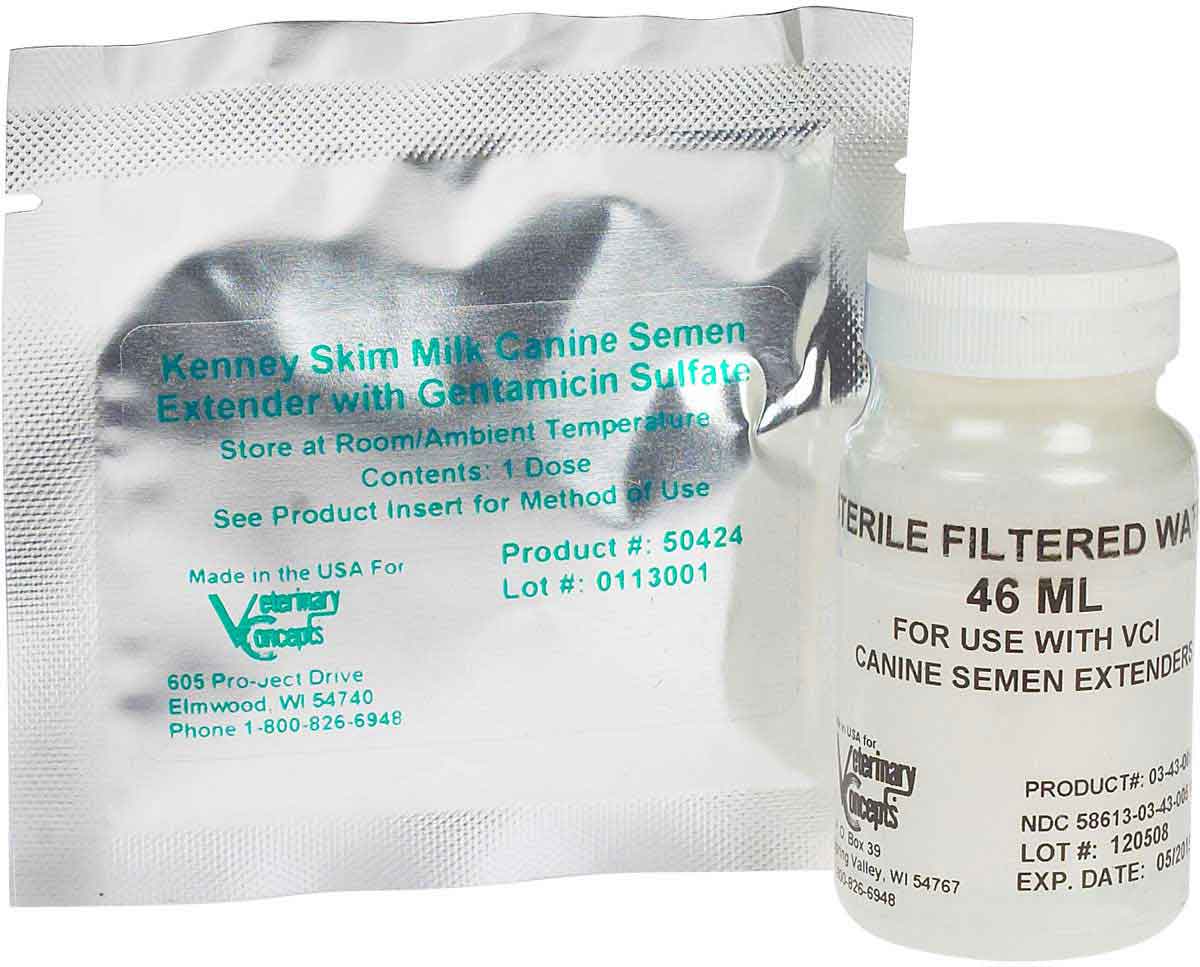 
The Kenney Skim Milk Canine Semen Extender Kit allows you to store cooled semen for use up to 48 hours after collection. Please state which one you require20ml of semen extender provided Easy to follow instructions Tried and tested successfully against the leading semen extendersCondition is New. New improved formula Canex canine semen extender with or without egg yolk added. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
